New Study Suggests Vaccination Makes You More Susceptible to COVID-19 in the Days After the Jab
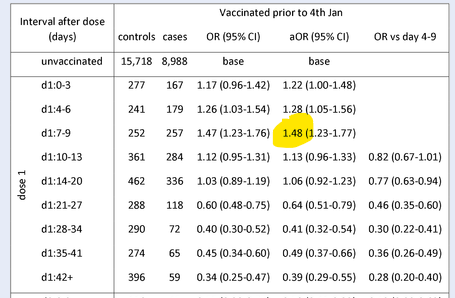
Lockdown Sceptics reported last week on two studies that had found an increase in COVID-19 infection risk in the week after the first vaccine dose. A PHE study found a 48% increase in infection risk in the over-80s group 4-9 days after receiving the first dose of the Pfizer vaccine (see table below). And the American FDA Emergency Use Authorisation for the Pfizer vaccine found 40% higher “suspected COVID” in the first week after vaccination compared to the control group. As a potential explanation, Lockdown Sceptics noted that in trials the Pfizer vaccine was found to suppress lymphocyte count in the first few days after treatment (see chart above), potentially increasing susceptibility.
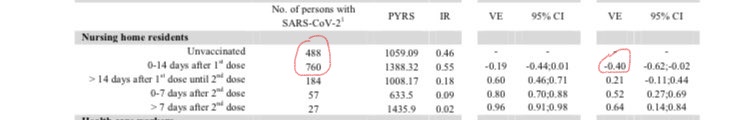
Now a new paper from Denmark has made a similar finding. Tracking every vaccination given to nursing home residents, it finds a 40% increase in infection risk in the 14 days following the first Pfizer dose (see table below). (Ultimately, it finds a 64% vaccine efficacy for nursing home residents a week after the second dose.)
Does this explain why there have been numerous reports of care home outbreaks shortly following vaccination? Could this be why Dr Hervé Seligmann found an elevated death rate among vaccinated people in Israel?
Is anyone in Government or the MHRA asking these questions, if only to rule out any problems?
Personally, I would be reassured to see the overall mortality rate of vaccinated and unvaccinated people over time. I cannot see why it is not routine to collect and publish this data, though it appears it is not. This would be the most straightforward way of establishing whether there is a potential problem that needs addressing in relation to short-term vaccine safety or there is not.
Will the Government publish this data to set our minds at ease?

To join in with the discussion please make a donation to The Daily Sceptic.
Profanity and abuse will be removed and may lead to a permanent ban.
Government publish data which will undermine their strategy? I think not.
Assuming the vertical bars on each entry in the lymphocyte versus day/dose graph represent a 95% confidence interval, then there is no statistically significant difference between the placebo and all day/drug dose cases, with one exception. For the Dose 1/Day 1-3 instance, there is no overlap between the 95% confidence intervals for the placebo and the 100ug dose. This indicates a statistically significant worsening for high doses shortly lafter administration. Everywhere else, placebo and dose values overlap, hence no statistically significant difference between them. Save the money and have a placebo stab.
Exactly what many commentators have been saying for weeks now, but meanwhile being dismissed as anti vax cranks.
Two huge questions :
– Informed consent. Are those getting the vaccine informed of the increased risk of infection and death?
– Would 60% efficacy and deaths related to the vaccine have been a block on approval in November? Especially given the group most affected are also the most vulnerable?
This is incredibly sketchy territory for anyone involved
This temporary Authorisation under Regulation 174 permits the supply to and by the Crown of COVID-19 Vaccine AstraZeneca, based on the safety, quality and efficacy data submitted by AstraZeneca to MHRA in the period from 24/09/2020 to 29/12/2020 December 2020.
This authorisation is not a marketing authorisation for the purposes of Part 5 of the HMRs or Chapter 4 of Title III to the 2001 Directive. (Gov.UK)
When those asking questions are blown off there is something nefarious afoot.